Cosmetics.
Helping you fast-track your product’s entry into the European market, the strictest market in the world, effortlessly
All cosmetic products sold in Europe must comply with the European regulation for placing cosmetic products on the European market.
As a cosmetics manufacturer, you are required to follow a 6-STEP process to register your product, under EU Cosmetics Regulation (EC) No. 1223/2009.
The regulation legally requires you to designate a “Responsible Person.“ That’s where Toxi Plan comes in! Our expert toxicologists and eco-toxicologists ensure your product’s regulatory compliance from STEPS 1 through 6.
The 6-STEP Cosmetic Registration Process.
The regulatory process consists of 6 mandatory steps.
We know it’s complicated, so we’ve simplified it for you!
“RESPONSIBLE PERSON“ (RP)
An RP is a legal or natural person based in the EU, like Toxi Plan toxicologists, who will act as your regulatory compliance representative, before your product can be placed on the shelves.
PRE-EVALUATION
This step allows you to evaluate your formula’s compliance, identify required testing and assess technical documentation.
COSMETICS PRODUCT SAFETY REPORT (CPSR)
The CPSR is a two-part report that confirms your product is safe. Part A: provides cosmetic product safety information and Part B: confirms safety assessments were completed and in compliance.
PRODUCT INFORMATION FILE (PIF)
Confirms the quality, safety and efficacy of a cosmetic product. Includes product description, the CSPR, GMP-compliant manufacturing methods, technical data supporting all claims, and data relating to animal testing. Must be updated on a regular basis.
LABELS AND CLAIMS VERIFICATION
A label and claims review ensures your packaging’s regulatory compliance. An important step before investing in marketing and design of your product.
COSMETIC PRODUCT NOTIFICATION (CPNP)
Once steps 2-5 are complete, your RP must submit your product file through the EU’s Cosmetic Products Notification Portal for registration.
Our Services.
Toxi Plan offers customizable toxicology, eco-toxicology and regulatory services to meet your market entry and sustainability needs.
Our expert toxicologists and eco-toxicologists ensure your product’s regulatory compliance from STEPS 1 through 6.
Our services go the extra mile! We incorporate eToxiSafe, a revolutionary new environmental impact assessment tool in our approach.
Sustainability is at the core of our business. We strongly believe it is the future of business and the only way to future-proof your success.
Our patented tool, the eToxiSafe calculator evaluates and predicts your formula’s environmental impact.
Our eco-labeling and certification service increases your competitive advantage and builds customer loyalty.
Our innovative and patented approach
Toxicology & Regulatory
(*Fulfils the regulatory 6-Steps)
-
Toxi Plan offers toxicological and regulatory consulting services to help you establish a customized registration plan.
This step allows you to evaluate your formula’s compliance, identify required testing and assess technical documentation.
-
Toxi Plan performs an toxicological for each INCI used in your product. We use it to set a "safe dose" for human use based on data collected from a scientific literature review.
Safety margins ensure that the safe dose will not be exceeded when the product is used.
-
The CPSR is a two-part report that confirms your product is safe for human use. Part A: provides cosmetic product safety information and Part B: confirms safety assessments were completed and in compliance.
-
Toxi Plan prepares your product’s PIF. It serves to confirm the quality, safety and efficacy of a cosmetic product. It includes product description, the CSPR, GMP-compliant manufacturing methods, technical data supporting all claims, and data relating to animal testing.
This is a mandatory component of the regulatory file. -
A label and claims review ensures your packaging’s regulatory compliance. An important step before investing in marketing and design of your product.
-
Toxi Plan will submit your product file through the EU’s Cosmetic Products Notification Portal for registration.
-
Toxi Plan prepares a Cosmetovigilance Report, a post-market surveillance that consists of collecting, assessing and monitoring undesirable effects of your product on human health and safety.
Eco-Toxicology & Regulatory
Ensure your product’s commitment to sustainability. It is the future of business and the only way to future-proof your success.
1. INCI Eco-Toxicological Profile and Safety Margin Calculation
2. eToxiSafe EcoToxImpact Assessment
3. Eco-Toxicity Laboratory Testing
4. eToxiSafe Environmental Compliance Report
5. eToxiSafe® Eco-Labeling Service
Cosmetics is our #1 business sector!
-
Shampoos, Conditioners, Hair Masks, Oils and Serums, Hair Dyes, etc.
-
Shower Gels, Deodorants, Bath Salts, Soaps, Toothpastes, Mouthwash, etc.
-
Mask, Scrubs, Serums, Oils, Face, Body and Hand Lotions, Lip Balms, Cleansers, Solar Products, etc.
-
Makeup, Perfumes and Colognes, Nail Polishes, etc.
Spotlight
Discover a selection of products expertly evaluated by our toxicologists!
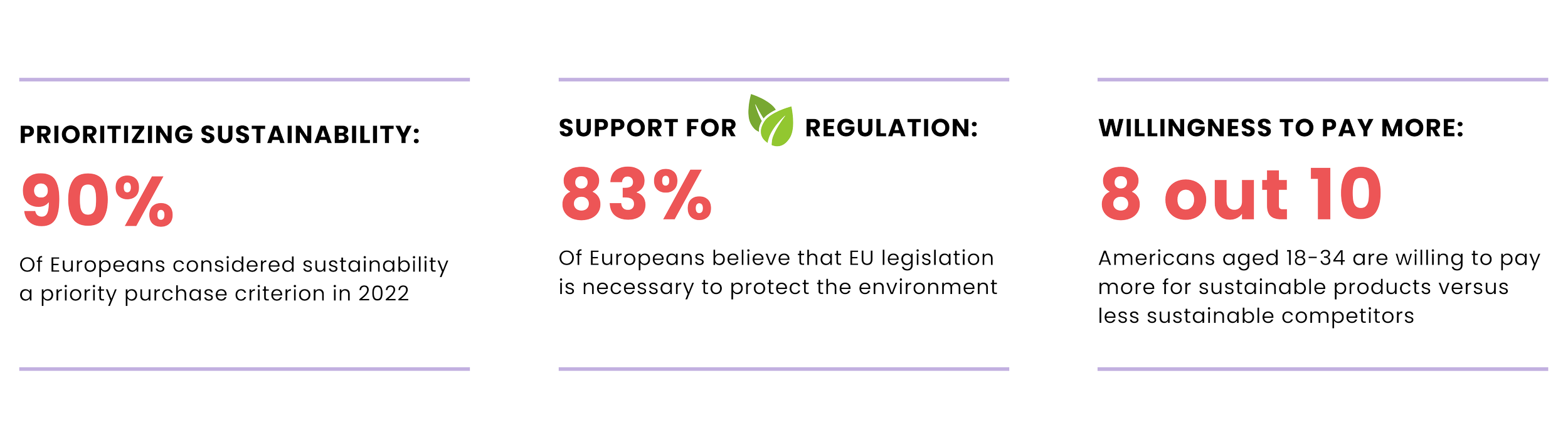
Why is sustainability the Future of Business?
Over the last decade, trends such as conscious consumerism, clean and green beauty combined with consumer calls to eliminate microplastics, for example, have put an increased focus on the industry’s impact on our planet. The pandemic has further drawn a focus on the importance of sustainability, driving awareness, and the need to take action.
- Koziani Zulkifli, Exhibitions Director, in-cosmetics Global
Get the lowdown on our services - download our brochure for easy access anytime!
Let’s work together.
Interested in working together? Fill out some info and we will be in touch shortly! We can't wait to hear from you!